A Deep Overview of Bioluminescence Imaging (BLI)
by Katharine Martin

by Katharine Martin
Bioluminescence imaging (BLI) has become an important
research tool for understanding biological processes
in vivo and in
real-time. Some of the immediate benefits of bioluminescence imaging is the low
signal-to-noise ratio (SNR), high sensitivity, and the ability for processes to
investigate
in vivo without harming the animal being studied. Here, we
detail what bioluminescence imaging is, how subjects are imaged, information
about equipment, substrate delivery and more.
What is bioluminescence imaging (BLI)?
A simple overview of the bioluminescent reaction
How is bioluminescence imaged?
Advances in BLI instrumentation
Different Bioluminescence Imaging Methods
BRET – Bioluminescence resonance energy transfer
Multi-reporter bioluminescent imaging
Steady-state bioluminescence imaging
Substrate delivery techniques in bioluminescence imaging
How does my animal model express luciferase for bioluminescence imaging?
Transgenic models in bioluminescence imaging
Mouse xenograft for bioluminescence imaging
Humanized tumor xenograft mice for bioluminescence imaging
Genetically engineered mice (GEM) for bioluminescence imaging
What applications use bioluminescence imaging (BLI)?
Bioluminescence imaging allows researchers to study biological processes in vivo and in real-time without being invasive or sacrificing the animal of study. BLI harnesses the concepts of bioluminescence, using the luciferase-luciferin system as a reporter for biological events at the molecular level.
Unlike fluorescence, BLI doesn’t rely on incident light – or light that shines on the subject, and therefore does not cause phototoxicity. Instead, BLI relies on the bioluminescent reaction catalyzed by the enzyme luciferase.
Before diving into bioluminescence imaging, it will help to understand how the bioluminescent reaction works. The general reaction using firefly luciferase involves the conversion of the substrate d-luciferin into oxyluciferin by using luciferase as the catalyst. The resulting reaction releases energy in the form of light.
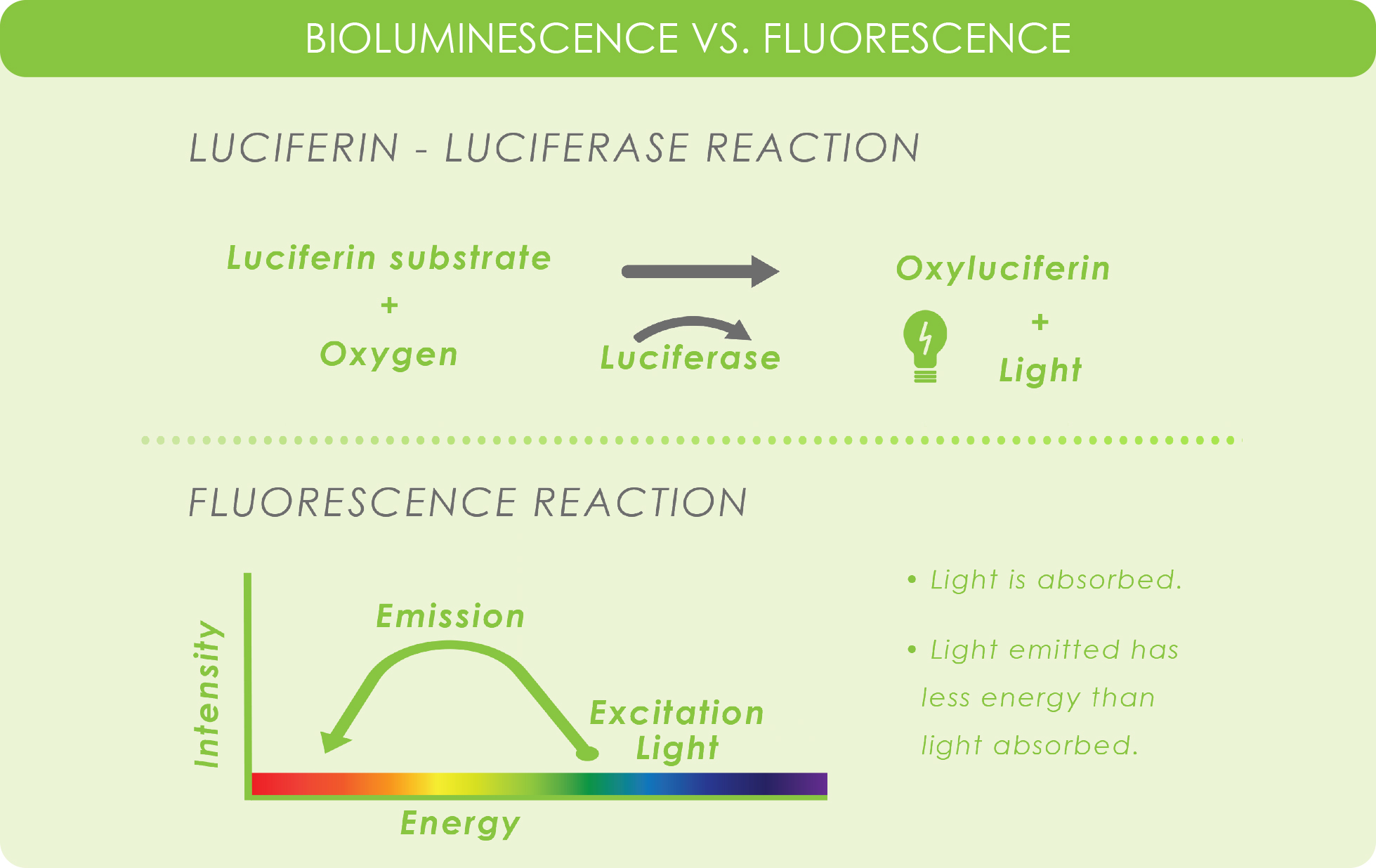
Usually, for in vivo research, scientists will use a transgenic organism such as a mouse or a rat that contains DNA encoding luciferase. When working with firefly luciferase, light won’t be emitted until its substrate, d-luciferin, is injected into the animal. Afterward, the animal is imaged and analyzed for light emission.
Bioluminescence is measured and analyzed using special imaging equipment and software programs. There are several available providers of bioluminescence imaging equipment on the market. These instruments have a large lightproof imaging compartment for the animal(s) being imaged, and use charged coupled device (CCD) cameras to capture the images (Close, Xu, Sayler, & Ripp, 2011).
The cameras are super cooled to -80°C in order to reduce thermal noise. Usually, these cameras will take two pictures. The first is a picture of the subject, and the second is the bioluminescent image which is then superimposed over the original image to accurately map light emission (Close, Xu, Sayler, & Ripp, 2011).
Different machines offer different features. For instance, some machines include gas anesthesia systems, heated stages and isolation chambers built in for better animal handling. There are different sized instruments available to accommodate different numbers of animals, benchtop machines for convenient use, and machines with enhanced cameras available for more sensitive detection.
While bioluminescence imaging provides significant advantages in molecular biology by enabling better understanding of molecular processes, there are limitations. Advances in imaging are therefore aimed at improving sensitivity, timing, and overall effectiveness.
One area of advancement pertains to the cameras being used in imaging systems. Some systems have begun using intensified CCD cameras (ICCD) or electron multiplying CCD cameras (EMCCD). These cameras image with extreme sensitivity in just milliseconds.
Other instruments have improved software allowing real-time imaging of conscious, moving animals. This helps with overall accuracy since anesthesia can sometimes interfere.
Though these improved systems help reduce some of the limitations of bioluminescence imaging, they are extremely expensive and still don’t completely solve some of the larger, looming concerns – the major one being how to deal with tissue depth, which restricts imaging to smaller animals (Close, Xu, Sayler, & Ripp, 2011).
There are different approaches to imaging that help researchers focus on specific processes such as tumor growth or shrinkage, expression events, co-expression events and more.
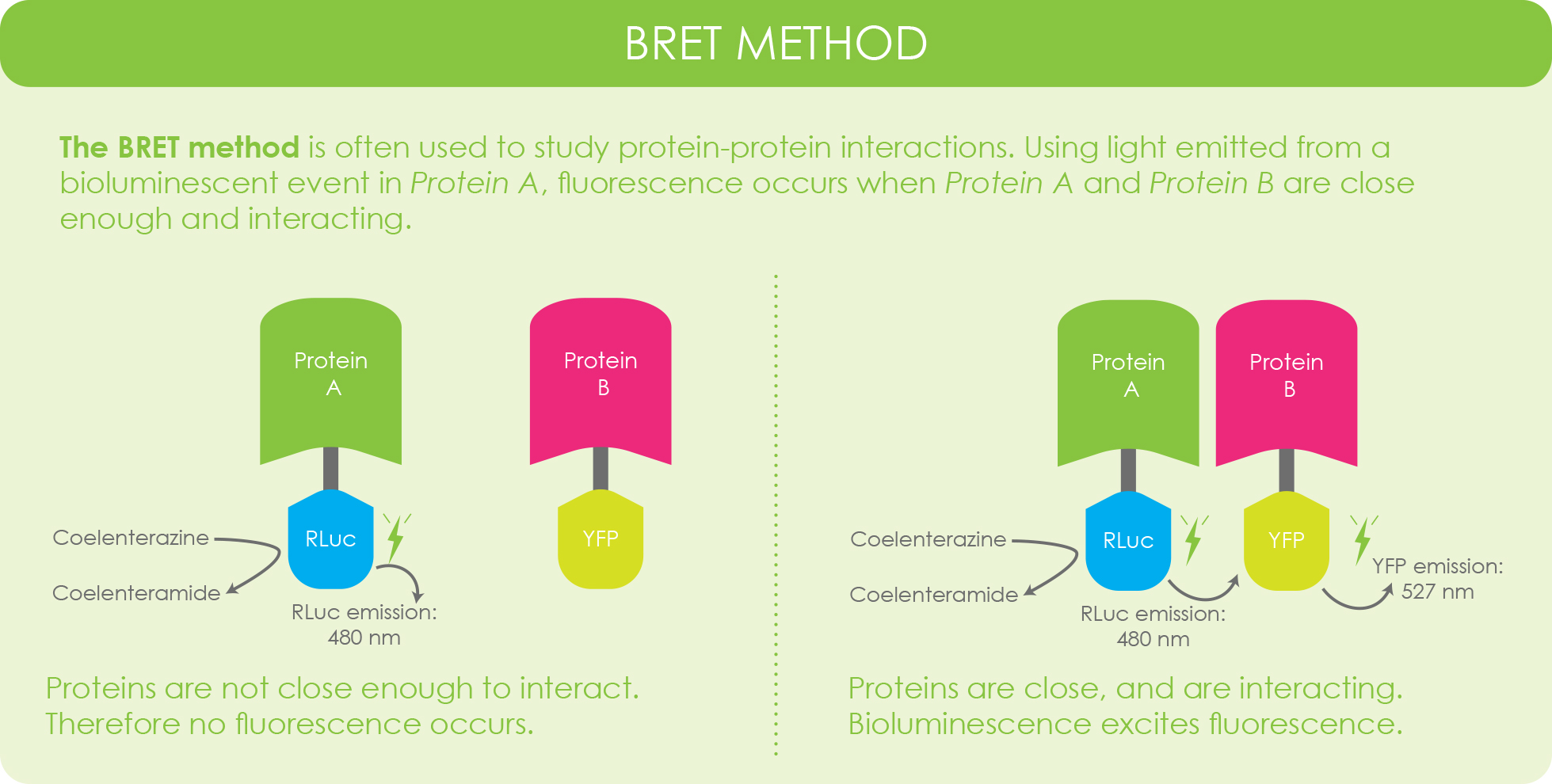
BRET, which stands for bioluminescence resonance energy transfer is a process that uses the light emission from a bioluminescent event to excite an associated fluorescent reporter.
This method has been extremely useful for studying protein-protein interactions, but it has also been used to help boost luminescent signaling (Close, Xu, Sayler, & Ripp, 2011).
Find a BRET protocol here.
Multi-reporter BLI uses two different luciferase reporters to study expression. Distinction between the two different reporting luciferases occurs through the different wavelength emissions. For example, firefly luciferase has a peak emission of 560 nm while Renilla reniformis has a peak emission of 480 nm.
Researchers use the BLI multi-reporter system to study codependent or interdependent protein expression. One example would be expression in circadian rhythm studies.
Using BLI rather than fluorescence enables an accurate perspective on events since there is no risk of excitation light impacting expression events (Close, Xu, Sayler, & Ripp, 2011).
Steady-state imaging is a very commonly used method that monitors the change in light emission over time. This process is especially helpful when studying tumor size changes. The benefit of using BLI is that the growth or shrinkage of a tumor can be monitored over time without the animal being sacrificed. Other processes might require time-series events using multiple animals at different stages of disease progression (Close, Xu, Sayler, & Ripp, 2011).

There are three different types of substrate delivery methods for BLI:
Researchers often use transgenic models for bioluminescence imaging. Usually these will either be transgenic mice or rats.

Transgenic mice’s genomes have been modified by the introduction of a sequence or artificial gene. Specifically, in bioluminescence imaging, transgenic animals are developed to express luciferase. But it’s not as simple as a mouse or rat expressing luciferase. In order to report on specific activity, researchers target certain promoters for luciferase expression. For example, to study p53 transcriptional activity, luciferase would be transcribed using two p53-dependent promoters: MDM2 and PUMA (Manni, de Latouliere, Gurtner, & Piaggio, 2019).
Researchers use xenograft mice expressing luciferase to study human cancers within mice. A xenograft mouse has had human cancer cells or tumors transplanted into the mouse’s organ of study (Richmond & Su, 2008). In order for foreign cells to be accepted, xenograft mice are immunocompromised.
The trouble with using immunocompromised mice is that the tumor is not in a system that mimics a human immune system or tumor microenvironment. Resulting studies, which may seem promising are not as effective in clinical settings (Morton, Bird, Refaeli, & Jimeno, 2016).
Humanized mice are produced when human hematopoietic stem cells and human tumor tissue is engrafted into immunodeficient mice strains. These models share more genes in common with the human genome and are therefore much easier to work with.
Furthermore, a xenograft of a human tumor cell using humanized mice has the benefits of better representing a human tumor microenvironment.
While humanized mice are a better model for tumor research, they are more expensive and more complicated (Richmond & Su, 2008).
Another approach to studying cancer progression and therapies is by using genetically engineered mice (GEM). Genetically engineered mice differ from xenograft mice in that genes of GEMs are altered. These are usually genes that pertain to malignancy and are changed so that they are deleted or overexpressed (Richmond & Su, 2008).
An advantage of using genetically engineered mice is that the tumor is being studied in an immunocompetent model. This means it will give researchers a better picture of the microenvironment. Another huge advantage is that tumor development can be studied earlier on compared to xenograft models.
While GEMs offer incredibly helpful advantages, there are disadvantages in their use such as their expense, time for validation and slow tumor development.
Bioluminescence imaging is foundational in many research applications. We’ve detailed how different luciferase systems are used within different applications. But in this section, we’ll provide a brief list of some BLI applications. Keep in mind, there are many more uses than what we present here (Badr & Tannous, 2011).
Ultimately, BLI assists researchers in a wide variety of disciplines including virology, immunology neurology, oncology and more.
Badr, C., & Tannous, B. (2011, December). Bioluminescence imaging: Progress and applications. Retrieved September 21, 2020, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC43149...
Close, D., Xu, T., Sayler, G., & Ripp, S. (2011). In vivo bioluminescent imaging (BLI): Noninvasive visualization and interrogation of biological processes in living animals. Retrieved September 21, 2020, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3274065/
Manni, I., De Latouliere, L., Gurtner, A., & Piaggio, G. (2019, February 25). Transgenic Animal Models to Visualize Cancer-Related Cellular Processes by Bioluminescence Imaging. Retrieved September 25, 2020, from https://www.frontiersin.org/articles/10.3389/fphar.2019.00235/full
Morton, J. J., Bird, G., Refaeli, Y., & Jimeno, A. (2016). Humanized mouse xenograft models: narrowing the tumor–microenvironment gap. Cancer research, 76(21), 6153-6158.
Nature Research. (n.d.). Bioluminescence imaging. Retrieved September 21, 2020, from https://www.nature.com/subjects/bioluminescence-imaging
Richmond, A., & Su, Y. (2008). Mouse xenograft models vs GEM models for human cancer therapeutics.
Sadikot, R., & Blackwell, T. (2005). Bioluminescence imaging. Retrieved September 21, 2020, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC27133...

A His-tag is a stretch of 6-10 histidine amino acids in a row that is used for affinity purification, protein detection, and biochemical assays. His-tags...

Competent cells such as DH5a, DH10B, and BL21 will maintain their transformation efficiency for at least a year with proper storage. It is important to...

Ni2+ ions give nickel agarose beads their characteristic blue color. This blue color can fade or disappear completely when loading his-tagged proteins onto the column....

Nickel agarose beads change from blue to a brown or black color when the nickel ions have been reduced from a Ni2+ to a Ni1+...
